 |
Polymorphism & Seeding
II. Case Studies |
 |
Polymorphism & Seeding
II. Case Studies |
Case Studies
Case 1. Cefadroxil (tradename Duricef etc.)
Background
Cefadroxil has been one of the most successful antibiotics of recent years and
is manufactured by Bristol Myers Squibb. Its patenting history provides a
classic case of the seeding and polymorphism situation described previously.
The Case
An early patent covered a form of cefadroxil (which might have been a
monohydrate or hemihydrate) named Micetich after the discoverer. The Micetich
form gave a poorly-resolved powder diffraction pattern that could not be
indexed. Much later Bouzard discovered a new form, which was much more highly
crystalline and easier to grow. This form is the subject of U.S. Patent
4,504,657 where the sole claim is as given below.
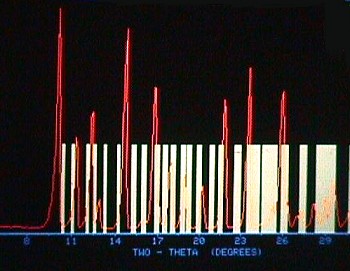
The sole claim of United States Patent 4,504,657 of Bouzard et al:
We Claim:
1. Crystalline 7-[D-α-amino-α-(p-hydroxyphenyl)acetamido]-3-methyl-3-cepham-4-carboxylic
acid monohydrate exhibiting essentially the following x-ray diffraction
properties:
| Line | Spacing d(Å) | Relative Intensity |
| 1 | 8.84 | 100 |
| 2 | 7.88 | 40 |
| 3 | 7.27 | 42 |
| 4 | 6.89 | 15 |
| 5 | 6.08 | 70 |
| 6 | 5.56 | 5 |
| 7 | 5.35 | 63 |
| 8 | 4.98 | 38 |
| 9 | 4.73 | 26 |
| 10 | 4.43 | 18 |
| 11 | 4.10 | 61 |
| 12 | 3.95 | 5 |
| 13 | 3.79 | 70 |
| 14 | 3.66 | 5 |
| 15 | 3.55 | 12 |
| 16 | 3.45 | 74 |
| 17 | 3.30 | 11 |
It is instructive to consider how precisely a powder diffraction pattern should be specified in a patent. If the claim is too broad (i.e., "We claim a crystalline substance having main powder diffraction peaks at approximately 8.84, 3.45 and 3.79 Å") then there is a danger that the claim would not be novel, because there are many previous substances that have main peaks at approximately those values. On the other hand if a claim is too exact it will certainly be novel (We claim crystalline 7-[D-α-amino-α-(p-hydroxyphenyl)acetamido]-3-methyl-3-cepham-4-carboxylic acid monohydrate with exactly the following x-ray diffraction properties... listing all 37 peaks), but there is a danger that this could make infringement impossible to prove, because it could be argued that unless each and every peak position and intensity in powder diffraction data from an alleged infringing material corresponds precisely to the quoted values it does not constitute an infringement. For instance, if the intensity of line 2 was 39 rather than 40 no infringement could be established! This is why the word essentially is used in the claim above; one would need to ask an expert in powder diffraction whether a given pattern does, or does not, exhibit essentially the same x-ray diffraction properties.
The crystallography
The the diffraction pattern from the new form was indexed to show that the
sample was pure and compared the patterns obtained from products from other
pharmaceutical companies who had tried to produce the Micetich form. In
laboratories which seeded with Bouzard's polymorph they were destined to fail.
On one occasion someone ran an experiment in a clean room with filters that
would eliminate anything from a bacterium upwards, but a typical bacterium is
several microns in size and a seed crystal could be hundreds of times smaller!
A brilliant scientific program developed at Oxford University by Dr. Nick Crouch carried out a series of experiments side-by-side, with one sample sealed from seed crystals and the other not. The seed-free experiments were reproducing the original Micetich conditions and produced the original polymorph. The open flasks (under otherwise identical conditions, but in a laboratory in which the Bouzard polymorph had been extensively produced) produced the new Bouzard polymorph, even without deliberate seeding. This program, supported by the testimony of Professor Sir Jack Baldwin and Dr. Bill Lipscomb on the organic synthesis and crystallography, and with powder diffraction as the key to characterising the phases, was a major step towards acceptance into United States Patent law of the need for provision for seeding and polymorphism cases, and hence into the patent laws of other countries.
Case 2. Ranitidine hydrochloride (tradename Zantac)
Background
Zantac has been the most successful anti-ulcer (H2-receptor
antagonist) drug to date, and until recently was the top-grossing drug of all
time. Glaxo (now Glaxo-Wellcome) discovered and patented ranitidine
hydrochloride, and this patent provides another case of the seeding and
polymorphism issue.
The Case
An early patent covered a range of related compounds including ranitidine
hydrochloride (and in common with most other such examples it was not until a
second polymorph appeared that powder diffraction became important). That form
only was scaled up and tested. Later on, as more and more work was being
carried out on ranitidine hydrochloride second polymorph appeared. The two
forms were then labelled 1 and 2. Form 2 grew much more quickly, and was
predominant in the presence of seeds. The subsequent patent for form 2 is U.S.
Patent 4,521,431. It describes how the x-ray data were collected and gives the
dnbsp&spacings and intensities. A characterisation was published in
Acta Cryst. 1990, C46, 1893-1896. "Structure of Ranitidine
Hydrochloride" by Ishida et al.
The crystallography
It was easy to show (and fairly predictable) that the structure found by Ishida
et al. was that of form 2, and hence the purity of form 2 samples was
established. Some claimed that form 1 was actually just some sort of impure
form 2, but from an indexing of the powder pattern from form 1 (an old sample
stored from the days before form 2 appeared) it was established that that was
also a pure single phase. One of the litigants attempted to take some
ranitidine hydrochloride form 2 back to ranitidine base and then follow the
final steps of the crystallization in the first (form 1) patent, but they got
form 2 back again because of seeding.
One must realise how strange and almost non-scientific these polymorphism and seeding cases can look to those with little or no experience in these matters. From the point of view of, say, a junior lawyer with little crystallographic knowledge and it might look as if a company such as Glaxo were somehow trying to extend their patent protection by sleight of hand, especially if the lawyer saw experiments attempting to replicate the form 1 patent, which actually produced form 2.
Many parties were interested in the question of whether there had been any form 2 in the form 1 samples dating from before the form 2 patent, because if so that patent might be invalid. Additionally, after the expiry of the form 1 patent, but while the form 2 patent was still in force, generic companies would be able to market form 1, but not form 2. Hence Glaxo would be interested to detect any form 2 in any samples produced by such companies. The problem would then become one of detection limits in quantitative analysis, as well as simple phase identification. This aspect is covered later in this section.
| © Copyright 1997-2006. Birkbeck College, University of London. | Author(s): Stephen Tarling |