 |
High Pressure |
 |
High Pressure |
High Pressure
Much of the information used in this topic has been supplied by Dr. Malcolm McMahon of the Department of Physics & Astronomy, The University of Edinburgh, UK, and to whom we are grateful.
The drive to subject materials to every increasing pressure has been fairly incessant since the second half of the 20th Century. We will first look at the historical background and then uncover the special role of powder diffraction in high pressure research:
Units
In this section we will refer to three units commonly used: an atmosphere which is the standard pressure exerted by our ambient air (e.g. ≈15 pounds per square inch or ≈1 Kg per square cm); a bar which to all extent and purpose is identical to the atmosphere; and a Gpa which is 10000 bars. Typical high pressure milestones are 100 kbars (100000 bars) and a Mbar (million bars or 100 Gpa).
Why High Pressure?
As already stated, the urge to pressurise materials has always been there and a very identifiable High Pressure Community now exists worldwide. Earlier the developments were left to a few enthusiasts. Of these the father is undoubtedly Percy W.Bridgman, 1882-1961, of whom it was said would squeeze just about anything he could lay his hands on. No material seemed to escape his attention and his published work included such all-embracing titles as "Rough compressions of 177 substances to 40000 kg.cm− 2"! He duly received the Nobel prize in 1946 for his prodigious work.
Much of the early drive in high pressure research was concerned with the holy grail of trying to make synthetic diamonds - even Bridgman himself occasionally became diverted into this quest. It was universally acknowledged that to attain synthetic diamonds one would require a combination of both high pressure and temperature. The race was effectively won in 1955 by a team, including Herbert Strong and Tracy Hall, from the General Electric Co. in USA by pressurising mixtures of carbon and metal up to an estimated 100 kbar (100000 atmospheres) at 1600°C for 38 minutes; incidentally another, Swedish, group had actually succeeded 2 years earlier but failed to publish the results (an object lesson here perhaps!); also there is a very nice read of the synthetic diamond story to be found in "the DIAMOND makers" by Robert M.Hazen, Cambridge University Press, 1999.
Although that particular motivation has now passed, the use of high pressure to synthesise new materials continues unabated. An interesting historical example of high pressure synthesis involved another USA scientist, Loring Coes, who in 1953 (publication date) squeezed quartz at 35000 atmospheres and 800°C to form a new dense silica phase which was later named "coesite". This result caught the imagination of scientists from the Geophysical Laboratories at Washington who realised that such results implied the mineralogy inside deep earth was very different and that here was a way to create these minerals within the laboratory; incidentally, Coes' synthetic silica was actually found in natural form a few years later by a geologist, Edward Chao, while examining the products of meteor collision at Meteor Crater, Arizona. Today such geologists (Earth and Planetary science) constitute a large fraction of the high pressure community. The following is typical of the sort of diagrams that are used in this context:
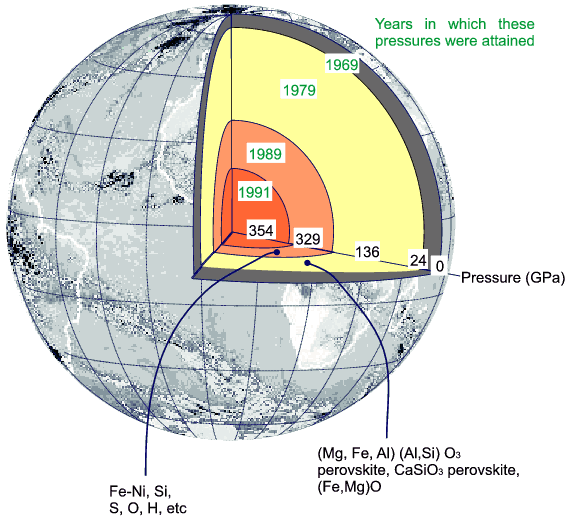
Why powders?
Structure solution and analysis from single crystal diffraction is much easier than from powder diffraction. However since under pressure materials are generally compressible and prone to unpredictable high order phase transitions (see the following schematic figure of a typical pressure, P, versus volume change [V/V0]) which can bring about a
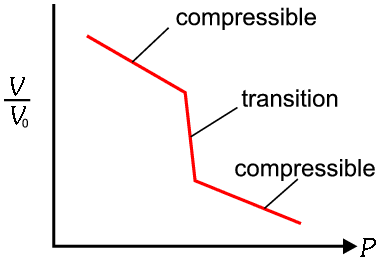
|
© Copyright 1997-2006.
Birkbeck College, University of London.
|
Author(s):
Paul Barnes Simon Jacques Martin Vickers |